|
This is the first period of the periodic table that involves d-block elements, 3d sub-shell in the process of completion for d-block. The last element Kr belongs to Nobel gases. Next 10 elements are from d-block and next six elements are from p-block. F-block elements are counted in the sixth and seventh periods. Elements of fourth period in modern periodic table. The sixth period and seventh period each have 32 elements. Periodic Table: Subject: Chemistry: Class: Class 11: Answer Type: Video solution: 3: Upvotes: 332: Avg. The fourth period and fifth period each have 18 elements. of elements in a period is decided by the possible orbitals as following (based on Aufbals rule). The second period and third period each have 8 elements. The first period has 2 elements, hydrogen and helium. All elements in a row have the same number of electron shells. 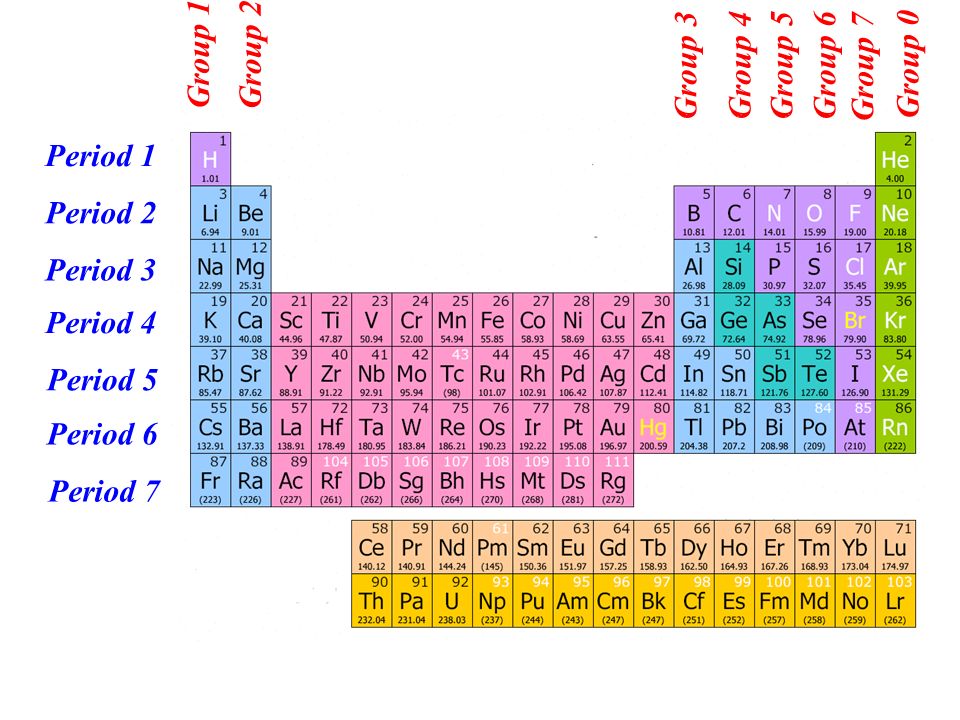
the proton number increases by one from one element to the next. Table shows the changes in the proton numbers and number of valence electrons when going across Period 2. The Periodic Table consists of seven periods, from Period 1 to Period 7. Whilst they are not based on the chemical reactivity they do share an interesting feature: electron shells. Each horizontal row of elements in the Periodic Table is known as a period. 
The smaller atoms are on the left and the larger ones are on the right because the atoms have more protons and electrons. A period in the periodic table is a row of chemical elements. Periods are the rows of the periodic table. 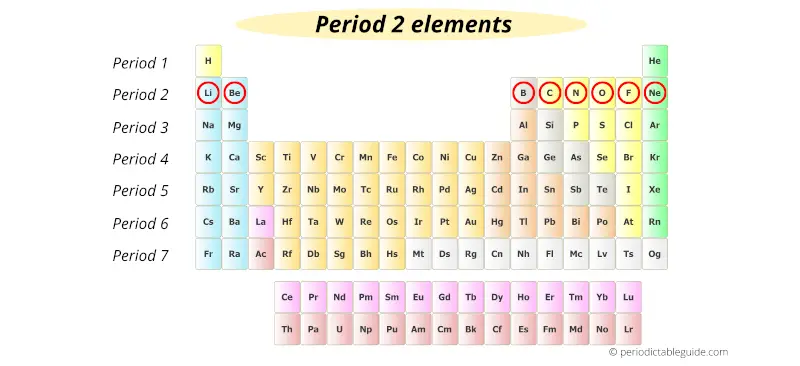
Period 2 elements have electrons in the first two electron shells.The first electron shell is always full. The elements in the same period are very metallic on the left and nonmetallic on the right. A period 2 element is an element in the second period (row) of the periodic table.Period 2 has eight elements: lithium, beryllium, boron, carbon, nitrogen, oxygen, fluorine and neon.Their atomic numbers go from 3 to 10. This means its atomic number is one number higher. Each elements in the same period has one more proton than the one to its left. This means they react with other elements in similar ways. A period in the periodic table is any horizontal row of elements. The formulation of the periodic table began during the. Elements in the same column are in the same group. Successive periods down the table correspond to atoms with a more electron-rich core of inner shells. This means they have similar physical properties, such as how well they bend or conduct electricity. Elements in the same row are in the same period. These electrons screen or shield the outer electrons from the nuclear charge.A period in the periodic table is any horizontal row of elements.Įach elements in the same period has one more proton than the one to its left. On the periodic table, elements are listed in order of increasing atomic number. This is because while the number of electrons increases down the period, they only add to the same main energy level, and therefore do not. 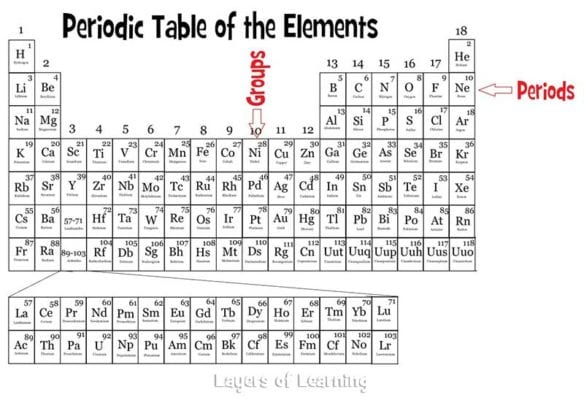
For example, ionization energy, electronegativity, and of course atomic radius which we will discuss now. This is because of the screening effect of the filled inner electron levels. There are many trends on the periodic table. The greater attraction between the increased number of protons (increased nuclear charge) and electrons, pulls the electrons closer together, hence the smaller size.Īs you move down a group in the periodic table, the covalent radius increases. The covalent radius (a measure of how large individual atoms are) shows different trends if you are moving across a period or down a group.Ī comparison of the relative covalent radii of atoms is shown in the diagram below.Īcross a period from left to right, the covalent radius decreases.Īs you move from left to right across the periodic table, atoms have more electrons in their outer energy level and more protons in their nucleus. Explore the organization of elements on the Periodic Table and pay special attention to energy levels of elements that share periods in this interactive. Patterns and trends in the periodic tableĬhemists observe patterns in different properties of elements as they are arranged in the periodic table.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
